Specialty drugs, often oral agents and self-injectables, are characterized by high costs and the need for specialized manufacturing, distribution, storage, and clinical oversight.1 These drugs commonly address rare or serious conditions such as cancer. For Medicare, plans may add drugs to the specialty tier if their cost exceeds the threshold set by the Centers for Medicare and Medicaid Services (CMS), which is $950 per month for a 30-day equivalent ingredient cost.2,3 Due to the higher cost of specialty medications, payers use various utilization management tools as a framework to assess clinical appropriateness, safety, and cost-effectiveness. Prior authorization is an operational utilization management tool requiring pre-approval to confirm medical necessity and steer use toward guideline-concordant, cost-effective alternatives. Step therapy is another utilization management strategy for guiding treatment sequencing by trialing of first-line options before other therapies, which is enforced via prior authorization. Providers must document a lack of success or intolerability with therapies in earlier treatment lines prior to requesting approval for a therapy whose coverage includes a step therapy requirement.4
Some oncology stakeholders have shared their experience with prior authorizations. In February 2026, the Community Oncology Alliance published a survey of oncology professionals examining utilization management processes. Many respondents reported unfavorable perceptions across utilization management measures, with step therapy identified as increasingly time-intensive and complex to manage for both providers and patients over time.5
For life science companies, patient and provider experience is important for initial and continued use of innovative therapies. When starting patients on therapy is more burdensome than expected, it can contribute to negative experiences or perceptions. Brand teams can improve this experience if they can identify, define, and communicate accurate expectations for coverage requirements. Our focus here will be on some of the friction that may occur for antineoplastics in Medicare Part D, given that there are unique considerations between how coverage for Part B and Part D drugs is communicated. When manufacturers are intentional in designing the experience for initiation of therapy and communicating coverage requirements, they can help reduce this friction, even if prior authorization or step therapy requirements are in place.
Identifying oncology step therapy in Medicare Part D plans
CMS makes Public Use Files (PUFs) available that contain Medicare Part D formulary information, including information on the presence of prior authorization and step therapy requirements. The PUFs are also the underlying data source for Medicare Plan Finder, which beneficiaries can use to explore available plan options. Plan Finder is intended to help beneficiaries identify plans that include coverage for their medications; however, it may not surface the full story on requirements for coverage of a given drug. Plans often include step therapy requirements in formulary notes or prior authorization materials, consistent with CMS guidance and documentation requirements. When this is the case, the drug will be reported with only a prior authorization requirement, and the step therapy requirement will be absent.
Data snapshot: Medicare Part D utilization management requirements for oncology therapies
In Medicare, antineoplastics are a protected class under Medicare Part D, which requires plans to have these products on formulary.6 However, protected class status does not necessarily preclude the use of utilization management tools, which may affect how and when patients can access therapy. Figure 1 shows the results of an analysis of Medicare Part D formularies using the CMS PUFs. We reviewed the top 150 antineoplastics and selected 10 products with diversity across tumor types, indications, competition, and manufacturers. We calculated the percentage of Part D enrollees (e.g., patients) who have coverage with a step therapy requirement in the PUFs.7
Figure 1: Step therapy landscape in Medicare Part D prescription drug coverage
| # | Therapy | Percent of Part D formulary files showing prior authorization | Percent of Part D formulary files showing a step therapy requirement |
|---|---|---|---|
| 1 | Brukinsa | 99%+ | 0% |
| 2 | Cabometyx | 99%+ | 0% |
| 3 | Calquence | 99%+ | 0% |
| 4 | Iclusig | 99%+ | 0% |
| 5 | Koselugo | 99%+ | 0% |
| 6 | Lorbrena | 99%+ | 0% |
| 7 | Qinlock | 99%+ | 0% |
| 8 | Stivarga | 99%+ | 0% |
| 9 | Verzenio | 99%+ | 0% |
| 10 | Xtandi | 99%+ | 0% |
Source: Milliman’s Analysis of CMS January 2026 enrollment information
A reasonable interpretation from this analysis is that the antineoplastic agents in scope require prior authorization but do not require step therapy. Our review of prior authorization criteria found that this is often not the case.
Medicare Part D: Where is the mismatch in oncology step therapy reporting?
To explore further, we researched publicly available coverage policies and prior authorization criteria for the drugs listed above to identify trial and failure requirements and support a clearer understanding of how these requirements are described across public materials and coverage policies. Our review included formulary and prior authorization documents from various Part D plans. Our approach was not intended to be comprehensive or systematic, as the presence of a single policy suggests that CMS formulary reporting may not represent the actual existence of step therapy for these antineoplastic products.
The example in Figure 2 was taken from the coverage policy of a national Medicare Part D plan, but was blinded because the actual source of coverage is not relevant.9 Note the requirement for step‑through of at least one drug in the “Required Medication Information” section and in the “Prerequisite Therapy Required” row of the prior authorization table. This requirement is articulated within the broader clinical criteria of the coverage policy itself and may not be evident when reviewing formulary placement alone.
Figure 2: Example of coverage policy of a national Medicare Part plan
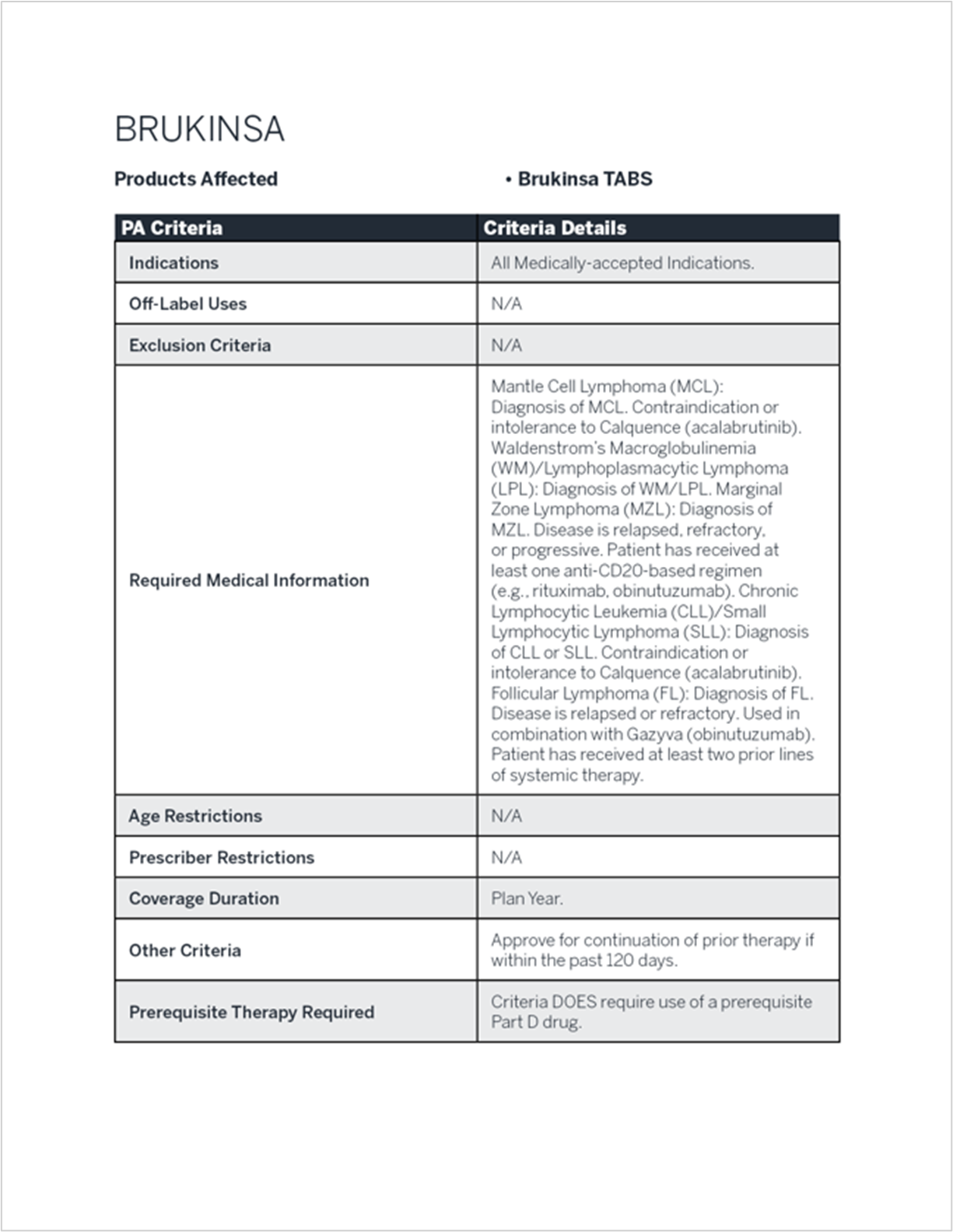
Figure 3 presents the results of our manual review of coverage documents limited to selected products. The step therapy landscape is complex, with requirements that may include progression through comparable branded therapies, chemotherapy, or hormonal treatments and that vary by indication. For our purposes, we have indicated step therapy when trial of another targeted therapy or prior use of chemotherapy is required.
Figure 3: Embedded step therapy criteria in Medicare Part D prescription drug coverage
| # | Therapy | Prior authorization required | Step therapy with at least one prior therapy identified through desk research* |
|---|---|---|---|
| 1 | Brukinsa | Yes | Yes |
| 2 | Cabometyx | Yes | Yes |
| 3 | Calquence | Yes | Yes |
| 4 | Iclusig | Yes | Yes |
| 5 | Koselugo | Yes | No |
| 6 | Lorbrena | Yes | Yes |
| 7 | Qinlock | Yes | Yes |
| 8 | Stivarga | Yes | Yes |
| 9 | Verzenio | Yes | Yes |
| 10 | Xtandi | Yes | No |
* “Yes” indicates at least one prior therapy is required for at least one indication, not necessarily all indications; “No” indicates no prior therapies are required.
The intended value of step therapy in oncology treatment pathways
Step therapy can play an appropriate role in care management and, in many cases, aligns with FDA labeling and clinical practice guidelines. Step therapy requirements may be absent when a therapy is FDA-labeled for first-line use, but this is not always the case with some of the examples above. Oncolytics have high toxicity, and the effectiveness of therapies can wane over time, necessitating careful selection of drugs based on many criteria. Utilization management, with clear parameters, can help providers deliver both cost-effective and guideline-concordant patient care. Step therapy can also serve as a cost-containment tool intended to steer providers to lower-cost therapies first, limit use of expensive drugs with uncertain incremental benefit, and help insurers manage prescription drug spending.10
Real-world impact of oncology step therapies on healthcare professionals and Medicare members
As noted above, step therapies are not consistently surfaced in public-facing plan tools and are often specified only in detailed prior authorization materials. As a result, a prescribed regimen may be out of sequence under the plan’s step therapy criteria at the time of submission. When this occurs, the patient’s healthcare team reviews the coverage criteria to identify a path forward and pursue authorization or exception pathways as appropriate. Depending on institutional workflows, prior authorizations, appeals, peer-to-peers, and additional documentation may fall to providers, pharmacists, non-clinical prior authorization staff, patient coordinators, nurse navigators, or financial counselors.11
Although utilization management is intended to support appropriate use of therapies, some healthcare professionals report that step therapy requirements in oncology may be associated with treatment delays, increased drug waste, a higher risk of avoidable complications, and disruptions to continuity of care with concerns of disease progression.12,13
Surveys and analyses from stakeholders examining patient-, provider-, and claims‑based perspectives indicate that prior authorization and related requirements are commonly encountered in oncology care, often involving direct patient or family engagement, extensive provider documentation, and appeals processes. Claims‑based analyses and physician-reported surveys further suggest that denials or rejections may influence delays in therapy initiation, continuation, and perceived clinical outcomes.14,15,16,17
Between 2010 and 2020, the prevalence of utilization management for oncology products covered under Medicare Part D increased and may continue to evolve as additional oral oncolytics and generic alternatives become available.18 Given this trend, there is an opportunity to further characterize how step therapy is implemented within the Part D formulary landscape and how it is understood across stakeholder groups.
Closing: How life sciences companies can cope with the challenges associated with Medicare Part D and oncology step therapy
Fundamentally, challenges in understanding the impact of step therapy requirements for oncology products in Medicare Part D stem from how coverage information is accessed and interpreted. Though the coverage documents referenced in this paper are publicly available, they are not always easy to find or understand.
Additional guidance may be needed to support consistency and understanding in how formulary coverage information is presented and interpreted. In other areas of healthcare, policy efforts have focused on improving the availability and clarity of information for patients. For example, the No Surprises Act of 2022 established protections to limit unexpected medical bills, while the Consolidated Appropriations Act of 2026 (H.R. 7148) includes provisions intended to increase insight into pharmacy benefit manager activities and rebate arrangements.
A potential consideration could be for CMS to highlight step therapy requirements with a consistent indicator across plan displays (e.g., Medicare Plan Finder and formulary files), such as “PA with ST,” to help healthcare professionals quickly identify when step therapy applies in addition to prior authorization.
Until the principles of improving navigation and understanding are applied to utilization management, specifically step therapy reporting in oncology in Medicare Part D, life science companies will need to create their own detailed coverage landscapes to help both with internal education and expectations from healthcare professionals and patients. In addition, life sciences companies should plan to take an active role in healthcare professional and patient communications to ensure that expectations are appropriate, coverage requirements are clear, and innovative therapies are as accessible as possible.
Our Milliman team has extensive experience navigating the complexity of oncology formulary coverage and can provide life sciences companies with a clearer understanding of how formulary placement and clinical criteria are implemented in practice.
1 Ismail, W.W., Witry, M.J., & Urmie, J.M. (2023). The association between cost sharing, prior authorization, and specialty drug utilization: A systematic review. Journal of Managed Care & Specialty Pharmacy, 29(5), 449-463. https://doi.org/10.18553/jmcp.2023.29.5.449.
3 Centers for Medicare and Medicaid Services. (2023, April 4). Final contract year (CY) 2024 Part D bidding instructions. Department of Health and Human Services. Retrieved April 6, 2026, from https://www.cms.gov/files/document/final-cy-2024-part-d-bidding-instructions.pdf.
4 Sachs, R.E., & Kyle, M.A. (2022). Step therapy's balancing act – Protecting patients while addressing high drug prices. New England Journal of Medicine. 386(10), 901-904. https://doi.org/10.1056/NEJMp2117582.
5 Community Oncology Alliance. (2026, February 10). COA 2026 utilization management survey. Retrieved April 6, 2026, from https://mycoa.communityoncology.org/publications/studies-and-reports/coa-2026-utilization-management-survey.
6 Legal Information Institute. (2025). 42 CFR § 423.120 – Access to covered Part D drugs. Cornell Law School. Retrieved April 6, 2026, from https://www.law.cornell.edu/cfr/text/42/423.120.
7 Centers for Medicare and Medicaid Services. (2026, January). Monthly enrollment by plan 2026 01. Retrieved April 6, 2026, from https://www.cms.gov/data-research/statistics-trends-and-reports/medicare-advantagepart-d-contract-and-enrollment-data/monthly-enrollment-plan/monthly-enrollment-plan-2026-01.
8 Centers for Medicare and Medicaid Services. (2026, January 8). Quarterly prescription drug plan formulary, pharmacy network, and pricing information. Retrieved April 6, 2026, from https://data.cms.gov/provider-summary-by-type-of-service/medicare-part-d-prescribers/quarterly-prescription-drug-plan-formulary-pharmacy-network-and-pricing-information.
9 The step therapy example given represents the policy of one Medicare payer. Across Medicare, Medicaid, and commercial channels, coverage policies can vary.
10 Sachs, R.E., & Kyle, M.A. (2022). Step therapy's balancing act – Protecting patients while addressing high drug prices. New England Journal of Medicine. 386(10), 901-904. https://doi.org/10.1056/NEJMp2117582.
11 National Community Oncology Dispensing Association. (2025, February 6). Preserving patient‑centric oncology care amid step therapy changes. Retrieved March 30, 2026, from https://www.ncoda.org/news/preserving-patient-centric-oncology-care-amid-step-therapy-changes/.
13 Lam, M., Olivier, T., Haslam, A., Tuia ,J., & Prasad, V. (2023). Cost of drug wastage from dose modification and discontinuation of oral anticancer drugs. JAMA Oncology. 9(9):1238-1244. https://jamanetwork.com/journals/jamaoncology/fullarticle/2807473.
14 American Society of Clinical Oncology. (2025, October 6). Prior authorization often places burden on patients with cancer, delays care. Retrieved April 6, 2026, from https://www.asco.org/about-asco/press-center/news-releases/prior-authorization-often-places-burden-patients-with-cancer-delays-care.
15 American Society of Clinical Oncology. (2022, November 22). Nearly all oncology providers report prior authorization causing delayed care, other patient harms. Retrieved April 6, 2026, from https://www.asco.org/news-initiatives/policy-news-analysis/nearly-all-oncology-providers-report-prior-authorization.
16 Thiesen, J., Glass, R., & Lamprecht, C. (2024, August). Access challenges in the cancer patient journey: How barriers to oral oncology affect patient initiation and persistency [White Paper]. IQVIA. Retrieved 6 April, 2026, from https://www.iqvia.com/-/media/iqvia/pdfs/us/white-paper/2024/iqvia-access-challenges-in-oncology-report-white-paper-2024.pdf.
17 American Medical Association. (2024, June 18). AMA survey indicates prior authorization wreaks havoc on patient care [Press Release]. Retrieved April 6, 2026, from https://www.ama-assn.org/press-center/ama-press-releases/ama-survey-indicates-prior-authorization-wreaks-havoc-patient-care.
18 Kyle, M.A., Dusetzina, S.B., & Keating, N.L. (2023). Utilization management trends in Medicare Part D oncology drugs, 2010-2020. Journal of the American Medical Association. 330(3), 278-280. https://doi.org/10.1001/jama.2023.10753.